Microscopy Images





Patients with crystalglobulinemia are usually aged 40-85 years with underlying plasma cell dyscrasia. In some cases, the increased precipitation of these crystals with cold exposure has led to use of the term cryocrystalglobulinemia.
Crystalglobulin-induced nephropathy often presents with severe acute kidney injury and patients with large vessel involvement can develop kidney infarction. This condition can be life threatening and associated with crystal precipitation in the vasculature of major organs and concurrent thrombosis. Other systemic manifestations include skin rash, polyarthralgia, vision impairment, gastrointestinal bleeding, and polyneuropathy.
Crystalglobulin-induced nephropathy has more commonly been associated with monoclonal gammopathy of renal significance and lymphoproliferative disorders rather than multiple myeloma.
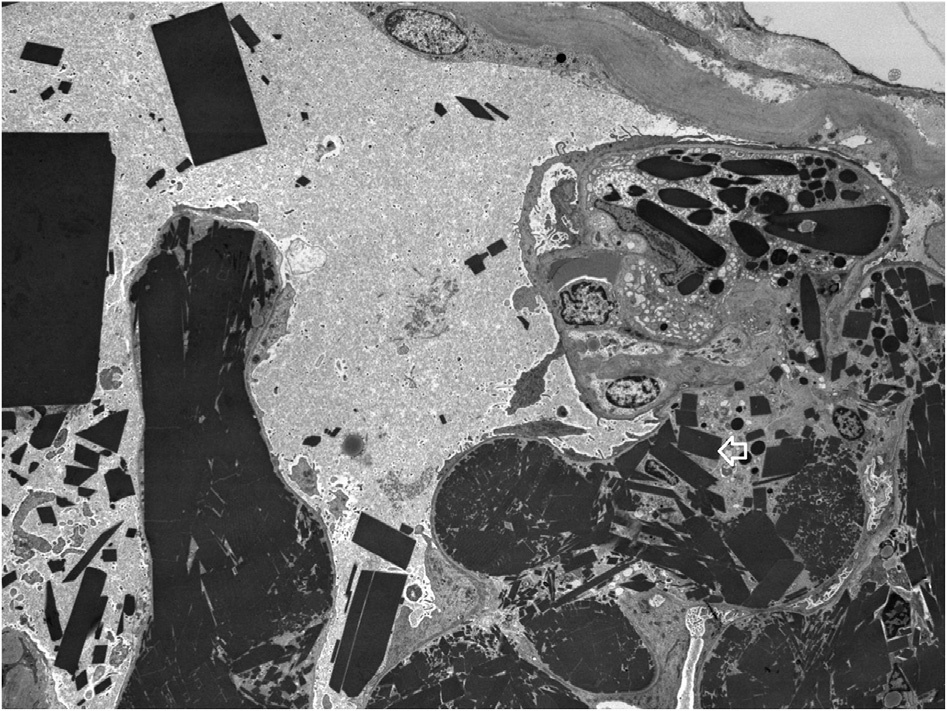
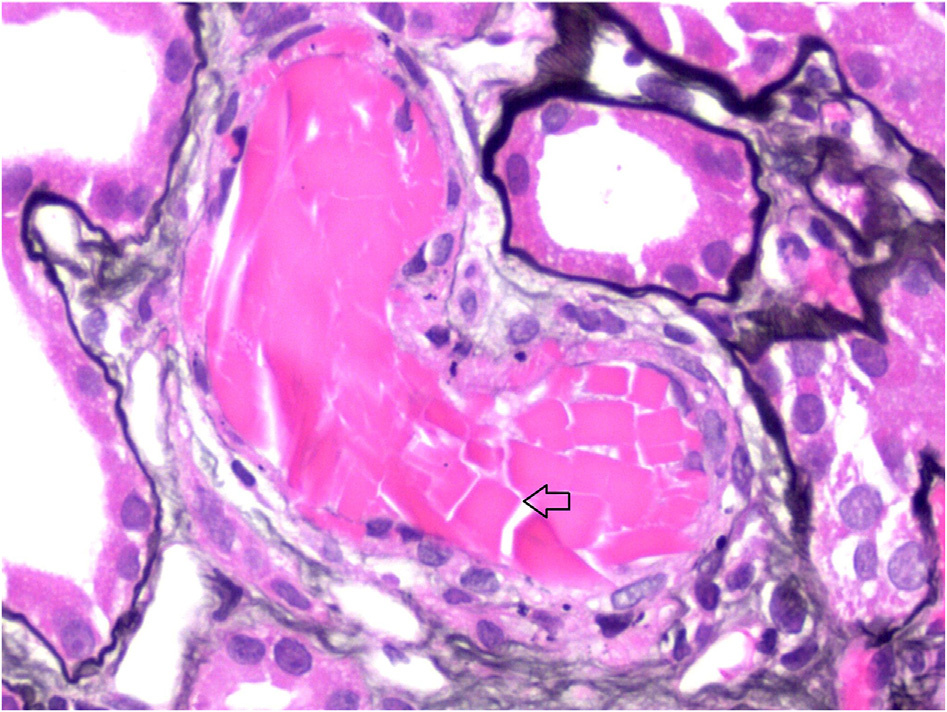
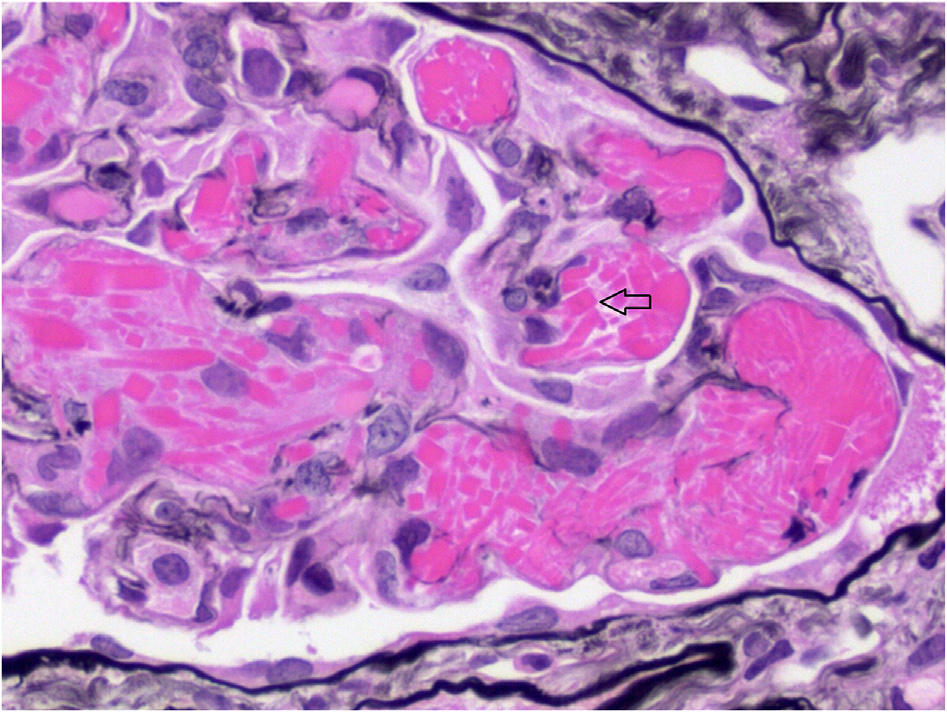
Light microscopy: Crystalglobulin-induced nephropathy results from extracellular deposition of monoclonal immunoglobulin precipitates within the renal microvasculature, including glomerular capillaries, and larger extraglomerular vessels. It is sometimes accompanied by numerous monocytes and neutrophils. These crystals can occlude vascular lumina mimicking thrombotic microangiopathy or induce arterial wallinflammation,resultingin vasculitisthatcanbeassociated with fibrin thrombi. The crystals are typically eosinophilic on hematoxylin and eosin staining, commonly periodic acid–Schiff (PAS) weak or negative (although they may be PAS positive as well), Jones negative, red on trichrome, and darkly stained with toluidine blue. The crystalline deposits may be needle shaped, rhomboidal, or irregular with sharp borders.
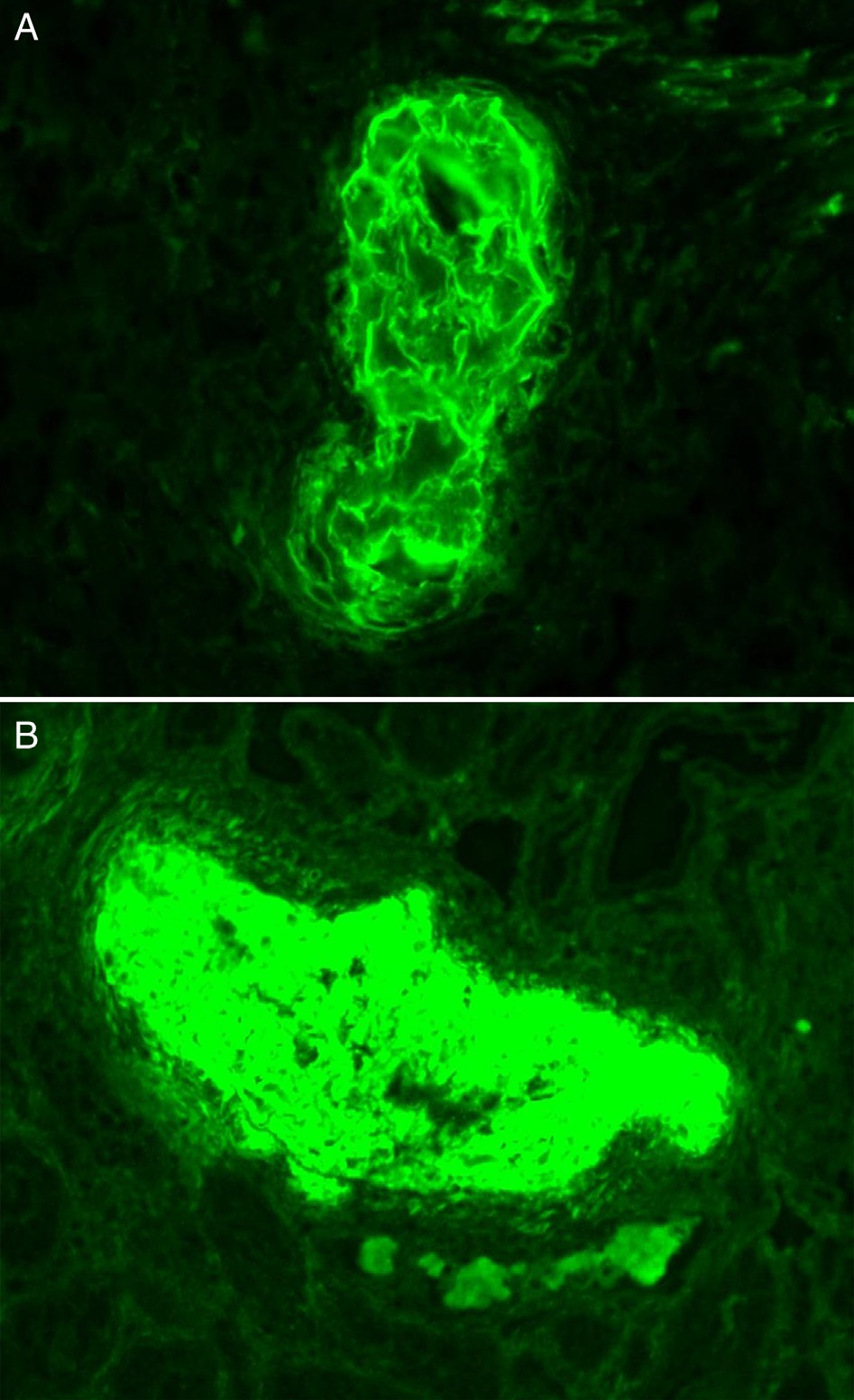
Immunofluorescence microscopy: The crystalline deposits are most commonly composed of IgGκ. Other paraproteins such as IgGλ, IgMκ, IgAλ, light chain alone, or polytypic IgG are also rarely reported. In about 50% of cases, immunofluorescence study on frozen tissue fails to detect the composition of crystalline deposits, and thus immunofluorescence study on pronase-digested paraffin-embedded tissue and/or immunoperoxidase study may be needed to confirm the monoclonal composition of the intravascular precipitates.
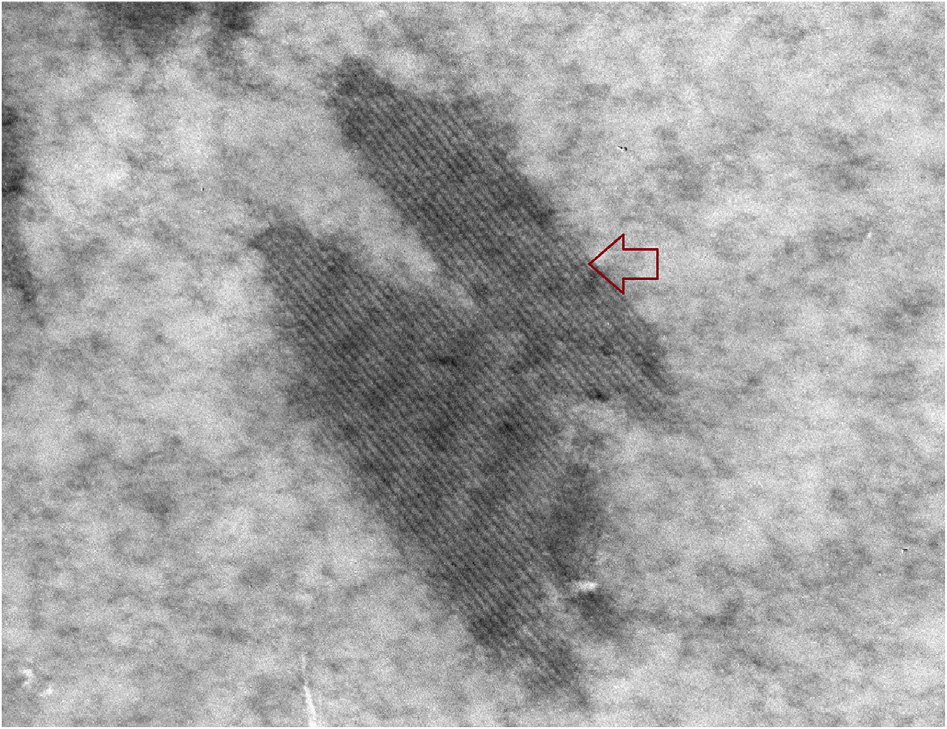
Electron microscopy: Deposits can be rhomboidal, rectangular, polyhedral, and/or needle shaped; electron dense; and crystalline, with sharp edges that may contain parallel linear array substructures, with periodicity of 14 nm and thickness of 6 nm described. The deposits are found in capillary lumens and/or arteries.
The mechanisms causing crystallization of the monoclonal protein are not well understood. Recent research suggests that this occurs because of FC/FC interaction of IgG-type monoclonal protein, possibly owing to abnormal glycosylation of the light chain or owing to interactions with albumin.
These mechanisms may be activated on cold exposure and/or with low flow in the circulation, analogous to the processes involved with cryoglobulin precipitation. The precipitation of crystalglobulin can cause endothelialinjury and result infibrin thrombi, which further increases ischemia and tissue injury.
Crystalglobulin-induced nephropathy should be distinguished from crystal-storing histiocytosis, cryoglobulinemic glomerulonephritis, and light chain proximal tubulopathy.
In crystal-storing histiocytosis, the crystals are mainly seen within interstitial histiocytes, but occasional crystals can be seen in proximal tubular cells and glomerular cells, particularly podocytes. Cryoglobulinemic glomerulonephritis also shows infiltrating monocytes within glomerular capillaries and can show PAS-positive plugs (cryo-plugs); however, these plugs lack crystalline appearance. Cryoglobulinemic glomerulonephritis usually shows mesangial and subendothelial deposits containing annular substructures. Light chain proximal tubulopathy is associated with intracellular rod- or rhomboid-shaped crystals within proximal tubular cells but not within the vascular lumina.
Article Information Authors’ Full Names and Academic Degrees: Paisit Paueksakon, MD, Behzad Najafian, MD, Shreeram Akilesh, MD, PhD, Charles E.
Alpers, MD, and Agnes B. Fogo, MD.
Authors’ Affiliations: Department of Pathology, Microbiology, and Immunology, Vanderbilt University, Nashville, Tennessee (PP, ABF); and Department of Pathology, University of Washington, Seattle, Washington (BN, SA, CEA).